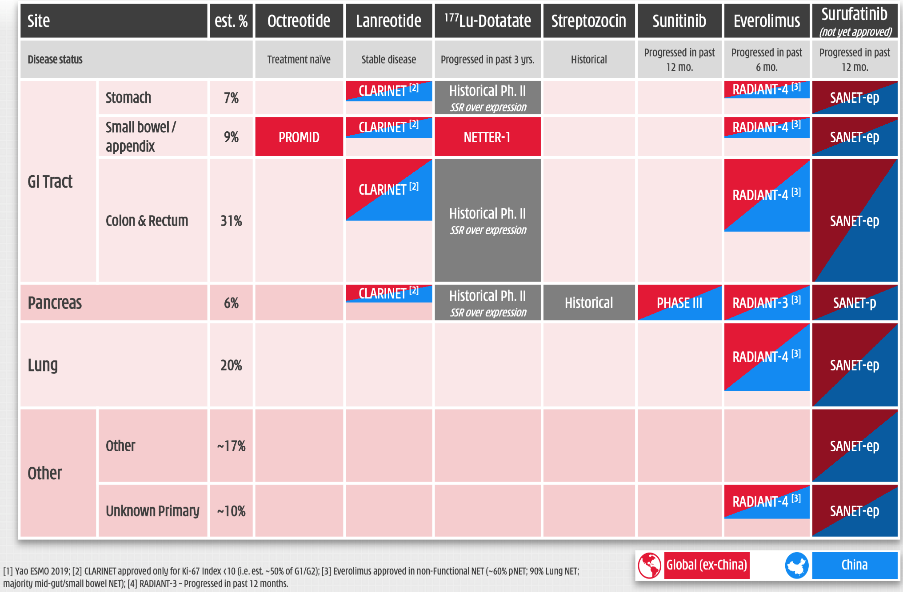
HCM reported consolidated group revenues of $204.9m in FY19 (FY18: $214.1m) and a group net loss of $106.0m (FY18: $74.8m). The depreciation of the Chinese renminbi versus the US dollar has affected top-line growth as reported in US dollars given the translation impact, as all revenues related to its China commercial platform (CP) business are generated in Chinese renminbi.
CP reported consolidated FY19 sales of $188.9m (+7% as reported, +11% CER; FY18: $176.5m), driven by the prescription drugs business, which now includes Elunate related manufacturing sales and royalties (HCM reported revenues of $10.8m in FY19 vs $3.6m in FY18) offsetting the impact of the termination of the Seroquel distribution agreement. Total consolidated net income from CP increased 9% to $47.4m (FY18: $43.4m). We forecast consolidated CP revenues of $196.6m in 2020 and $204.7m in 2021. The innovation platform (IP) reported consolidated revenues of $16.0m in FY19 compared to $37.6m in FY18, as FY18 benefited from the $13.5m Elunate approval related milestone received from LLY. In FY19, IP reported a net segment operating loss of $133.3m (FY18: $104.6m).
The profit before tax and equity in earnings of equity investees at group level reported a loss of $141.1m in FY19 (vs a loss of $86.7m in FY18). R&D expenses increased significantly to $138.2m in FY19 ($114.2m in FY18), reflecting investment throughout the portfolio, expansion of the US and international clinical and regulatory operations, and establishment of the China oncology commercial infrastructure. For FY20, HCM has guided to an adjusted non-GAAP IP segment operating loss of $180–210m and adjusted non-GAAP group net cash flow excluding financing activities of $140–160m.
We forecast R&D expenses to increase to $183.0m in 2020 and $210.0m in 2021 (on a reported GAAP basis), reflecting the substantial need for investment in the burgeoning clinical trial programmes across the IP division, including the increased investment in China and global trials plus the initiation of combination strategies across the portfolio. With the likely launch of surufatinib in China by end 2020 and the US in 2022, we expect sales and marketing expenses to accelerate significantly as HCM builds out its global commercial operations. We expect an increase in capex in 2021 to support investment in a new manufacturing facility: we forecast ~$42m per annum in 2021 and 2022.
We forecast net losses at group level of $163.4m in 2020 and $174.2m in 2021. HCM reported a strong cash position, with available cash resources of over $300m (at 31 December 2019) at group level (cash and cash equivalents and short-term investments of $217.2m, and unutilised bank borrowing facilities of $119.3m). Furthermore, HCM raised net proceeds of $110m from capital issuance in January 2020 and $100m (net) from the equity investment from General Atlantic. We believe the share price in 2021 will likely be higher than the exercise price of the warrant and thus forecast the additional $100m net proceeds is raised. Additionally, HCM’s non-consolidated joint ventures (Shanghai Hutchison Pharmaceuticals (SHPL) and Hutchison Baiyunshan (HBYS)) held $63.0m (at 31 December 2019). We note the JV Nutrition Science Partners (NSPL) has been acquired leading to a consolidated net cash inflow of $8.1m.
HCM recently announced that HBYS has come to an agreement with the Guangzhou government for the planned return of HBYS’s vacant land (HBYS Plot 2, a ~30,000 square metre site). HBYS will receive cash compensation of up to $95m in several stages over the next year as the transaction progresses to completion. We expect that ~40% of the compensation received will make its way to HCM via special dividends, which will be reinvested in the business.
Exhibit 19: Financial summary
|
US$'000s |
|
2017 |
2018 |
2019 |
2020e |
2021e |
Year end 31 December |
|
|
US GAAP |
US GAAP |
US GAAP |
US GAAP |
US GAAP |
PROFIT & LOSS |
|
|
|
|
|
|
|
Revenue |
|
|
241,203 |
214,109 |
204,890 |
216,750 |
283,912 |
Cost of Sales |
|
|
(175,820) |
(143,944) |
(160,152) |
(175,919) |
(196,866) |
Gross Profit |
|
|
65,383 |
70,165 |
44,738 |
40,831 |
87,046 |
Research and development |
|
|
(75,523) |
(114,161) |
(138,191) |
(183,000) |
(210,000) |
Other overheads |
|
|
(43,277) |
(48,645) |
(52,934) |
(60,021) |
(87,679) |
EBITDA |
|
|
(50,692) |
(88,975) |
(141,250) |
(194,454) |
(196,924) |
Operating Profit (before amort. and except.) |
|
|
(53,417) |
(92,641) |
(146,387) |
(202,190) |
(210,633) |
Intangible Amortisation |
|
|
0 |
0 |
0 |
0 |
0 |
Operating Profit |
|
|
(53,417) |
(92,641) |
(146,387) |
(202,190) |
(210,633) |
Net Interest |
|
|
(235) |
4,969 |
3,914 |
4,295 |
3,035 |
Exceptionals |
|
|
0 |
0 |
0 |
0 |
0 |
Profit Before Tax (norm) |
|
|
(53,536) |
(86,655) |
(141,106) |
(197,895) |
(207,597) |
Profit Before Tax (reported) |
|
|
(53,536) |
(86,655) |
(141,106) |
(197,895) |
(207,597) |
Tax |
|
|
(3,080) |
(3,964) |
(3,274) |
(3,300) |
(5,000) |
Equity investments, after tax |
|
|
33,653 |
19,333 |
40,700 |
42,769 |
43,418 |
Profit After Tax (norm) |
|
|
(22,963) |
(71,286) |
(103,680) |
(158,426) |
(169,179) |
Profit After Tax (reported) |
|
|
(22,963) |
(71,286) |
(103,680) |
(158,426) |
(169,179) |
Minority |
|
|
(3,774) |
(3,519) |
(2,345) |
(5,000) |
(5,000) |
Discontinued operations |
|
|
0 |
0 |
0 |
0 |
0 |
Net profit (norm) |
|
|
(26,737) |
(74,805) |
(106,025) |
(163,426) |
(174,179) |
Net profit (reported) |
|
|
(26,737) |
(74,805) |
(106,025) |
(163,426) |
(174,179) |
|
|
|
|
|
|
|
|
Average Number of Shares Outstanding (m) |
|
|
617.2 |
664.3 |
665.7 |
710.6 |
718.9 |
EPS - normalised (c) |
|
|
(4.3) |
(11.3) |
(15.9) |
(23.0) |
(24.2) |
EPS - normalised and fully diluted (c) |
|
|
(4.3) |
(11.3) |
(15.9) |
(23.0) |
(24.2) |
EPS - (reported) (c) |
|
|
(4.3) |
(11.3) |
(15.9) |
(23.0) |
(24.2) |
|
|
|
|
|
|
|
|
Average number of ADS outstanding (m) |
|
|
123.4 |
132.9 |
133.1 |
142.1 |
143.8 |
Earnings per ADS - normalised ($) |
|
|
(0.02) |
(0.06) |
(0.08) |
(0.11) |
(0.12) |
Earnings per ADS ($) |
|
|
(0.02) |
(0.06) |
(0.08) |
(0.11) |
(0.12) |
|
|
|
|
|
|
|
|
BALANCE SHEET |
|
|
|
|
|
|
|
Fixed Assets |
|
|
165,737 |
161,577 |
148,100 |
153,133 |
194,348 |
Intangible Assets |
|
|
3,738 |
3,533 |
3,387 |
3,000 |
2,315 |
Tangible Assets |
|
|
14,220 |
16,616 |
20,855 |
33,506 |
62,381 |
Investments |
|
|
147,779 |
141,428 |
123,858 |
116,627 |
129,653 |
Current Assets |
|
|
432,195 |
370,541 |
317,022 |
374,327 |
256,131 |
Stocks |
|
|
11,789 |
12,309 |
16,208 |
14,459 |
16,181 |
Debtors |
|
|
53,566 |
56,392 |
59,023 |
53,445 |
23,335 |
Cash |
|
|
85,265 |
86,036 |
121,157 |
281,800 |
191,992 |
St investments |
|
|
273,031 |
214,915 |
96,011 |
0 |
0 |
Other |
|
|
8,544 |
889 |
24,623 |
24,623 |
24,623 |
Current Liabilities |
|
|
(104,600) |
(85,479) |
(113,101) |
(125,870) |
(125,067) |
Creditors |
|
|
(25,344) |
(26,180) |
(25,789) |
(38,558) |
(37,755) |
Short term borrowings |
|
|
(29,987) |
0 |
0 |
0 |
0 |
Other |
|
|
(49,269) |
(59,299) |
(87,312) |
(87,312) |
(87,312) |
Long Term Liabilities |
|
|
(8,366) |
(34,383) |
(39,118) |
(39,118) |
(39,118) |
Long term borrowings |
|
|
0 |
(26,739) |
(26,818) |
(26,818) |
(26,818) |
Other long term liabilities |
|
|
(8,366) |
(7,644) |
(12,300) |
(12,300) |
(12,300) |
Net Assets |
|
|
484,966 |
412,256 |
312,903 |
362,472 |
286,294 |
Minority |
|
|
(23,233) |
(23,259) |
(24,891) |
(29,891) |
(34,891) |
Shareholder equity |
|
|
461,733 |
388,997 |
288,012 |
332,581 |
251,403 |
|
|
|
|
|
|
|
|
CASH FLOW |
|
|
|
|
|
|
|
Operating Cash Flow |
|
|
(8,943) |
(32,847) |
(80,912) |
(123,364) |
(140,910) |
Net Interest |
|
|
0 |
0 |
0 |
0 |
0 |
Tax |
|
|
0 |
0 |
0 |
0 |
0 |
Capex |
|
|
(5,019) |
(6,364) |
(8,565) |
(20,000) |
(41,898) |
Acquisitions/disposals |
|
|
0 |
0 |
8,689 |
0 |
0 |
Dividends |
|
|
(1,594) |
(1,282) |
(1,282) |
(2,000) |
(2,000) |
Equity financing and capital movements |
|
|
291,737 |
(2,322) |
(95) |
210,000 |
95,000 |
Other |
|
|
(255,761) |
50,116 |
118,904 |
96,006 |
0 |
Net Cash Flow |
|
|
20,420 |
7,301 |
36,739 |
160,643 |
(89,808) |
Opening net debt/(cash) |
|
|
(56,914) |
(328,309) |
(274,212) |
(190,350) |
(254,982) |
Increase/(decrease) in ST investments |
|
|
248,761 |
(58,116) |
(118,904) |
(96,011) |
0 |
Other |
|
|
2,214 |
(3,282) |
(1,697) |
0 |
0 |
Closing net debt/(cash) |
|
|
(328,309) |
(274,212) |
(190,350) |
(254,982) |
(165,174) |
Source: HCM accounts, Edison Investment Research
Contact details |
Revenue by geography |
Level 18, The Metropolis Tower
10 Metropolis Drive
Hung Hom, Kowloon
Hong Kong
+852 2121 8200
www.chi-med.com |
N/A |
Contact details |
Level 18, The Metropolis Tower
10 Metropolis Drive
Hung Hom, Kowloon
Hong Kong
+852 2121 8200
www.chi-med.com |
Revenue by geography |
N/A |
Management team |
|
Chairman: Simon To |
CEO: Christian Hogg |
Mr To has been a director since 2000 and an executive director and chairman of Hutchison China MediTech Limited since 2006. He is managing director of Hutchison Whampoa (China) Limited (Hutchison China) and has been with Hutchison China for over 39 years, building its business from a small trading company to a multi-billion-dollar investment group. Mr To’s career in China spans more than 44 years. He is the original founder of the China healthcare business of Hutchison Whampoa Limited (HWL) (currently a subsidiary of CK Hutchison Holdings Limited (CKHH)) and has been instrumental in the acquisitions made to date. He received a bachelor’s degree in mechanical engineering from Imperial College, London and a master in business administration from Stanford University’s Graduate School of Business. |
Mr Hogg has been the chief executive officer and an executive director of the company since 2006. He joined the business in 2000, as its first employee, and has since led all aspects of the creation, implementation and management of the company’s strategy, business and listings. This includes the establishment of the innovation platform, Hutchison MediPharma, which now comprises eight drug candidates that are being investigated in clinical studies around the world and a scientific team of about 500 people. Furthermore, Mr Hogg oversaw the acquisition and operational integration of assets that led to the formation of the company’s commercial platform, which manufactures, markets and distributes prescription drugs and consumer health products, covering an extensive network of hospitals across China. Prior to joining the company, Mr Hogg spent 10 years with P&G, starting in the United States in finance and then brand management in the laundry and cleaning products division. Mr Hogg then moved to China to manage P&G’s detergent business, followed by a move to Brussels to run P&G’s global bleach business. Mr Hogg received a bachelor’s degree in civil engineering from the University of Edinburgh and a master in business administration from the University of Tennessee. |
CFO: Johnny Cheng |
CSO: Weiguo Su |
Mr Cheng has been CFO since 2008. Prior to joining the company, Mr Cheng was vice president, finance of Bristol Myers Squibb in China and was a director of Sino-American Shanghai Squibb Pharmaceuticals Ltd. And Bristol-Myers Squibb (China) Investment Co. Ltd. in Shanghai between late 2006 and 2008. Mr Cheng started his career as an auditor with Price Waterhouse (currently PricewaterhouseCoopers) in Australia and then KPMG in Beijing before spending eight years with Nestlé China where he was in charge of a number of finance and control functions in various operations. Mr Cheng received a bachelor of economics, accounting major from the University of Adelaide and is a member of Chartered Accountants Australia and New Zealand. |
Dr Su has been the executive vice president and chief scientific officer of the company since 2012. Dr Su has headed all drug discovery and research since he joined the company, including master-minding the company’s scientific strategy, being a key leader of the Innovation Platform, and responsible for the discovery of each and every small molecule drug candidate in the company’s product pipeline. Prior to joining the company in 2005, Dr Su spent 15 years with the US research and development department of Pfizer, Inc. with his last position as director of the medicinal chemistry department. Dr Su received a bachelor of science degree in chemistry from Fudan University in Shanghai. He completed a PhD and post-doctoral fellowship in chemistry at Harvard University under the guidance of Nobel Laureate Professor EJ Corey. |
Management team |
Chairman: Simon To |
Mr To has been a director since 2000 and an executive director and chairman of Hutchison China MediTech Limited since 2006. He is managing director of Hutchison Whampoa (China) Limited (Hutchison China) and has been with Hutchison China for over 39 years, building its business from a small trading company to a multi-billion-dollar investment group. Mr To’s career in China spans more than 44 years. He is the original founder of the China healthcare business of Hutchison Whampoa Limited (HWL) (currently a subsidiary of CK Hutchison Holdings Limited (CKHH)) and has been instrumental in the acquisitions made to date. He received a bachelor’s degree in mechanical engineering from Imperial College, London and a master in business administration from Stanford University’s Graduate School of Business. |
CEO: Christian Hogg |
Mr Hogg has been the chief executive officer and an executive director of the company since 2006. He joined the business in 2000, as its first employee, and has since led all aspects of the creation, implementation and management of the company’s strategy, business and listings. This includes the establishment of the innovation platform, Hutchison MediPharma, which now comprises eight drug candidates that are being investigated in clinical studies around the world and a scientific team of about 500 people. Furthermore, Mr Hogg oversaw the acquisition and operational integration of assets that led to the formation of the company’s commercial platform, which manufactures, markets and distributes prescription drugs and consumer health products, covering an extensive network of hospitals across China. Prior to joining the company, Mr Hogg spent 10 years with P&G, starting in the United States in finance and then brand management in the laundry and cleaning products division. Mr Hogg then moved to China to manage P&G’s detergent business, followed by a move to Brussels to run P&G’s global bleach business. Mr Hogg received a bachelor’s degree in civil engineering from the University of Edinburgh and a master in business administration from the University of Tennessee. |
CFO: Johnny Cheng |
Mr Cheng has been CFO since 2008. Prior to joining the company, Mr Cheng was vice president, finance of Bristol Myers Squibb in China and was a director of Sino-American Shanghai Squibb Pharmaceuticals Ltd. And Bristol-Myers Squibb (China) Investment Co. Ltd. in Shanghai between late 2006 and 2008. Mr Cheng started his career as an auditor with Price Waterhouse (currently PricewaterhouseCoopers) in Australia and then KPMG in Beijing before spending eight years with Nestlé China where he was in charge of a number of finance and control functions in various operations. Mr Cheng received a bachelor of economics, accounting major from the University of Adelaide and is a member of Chartered Accountants Australia and New Zealand. |
CSO: Weiguo Su |
Dr Su has been the executive vice president and chief scientific officer of the company since 2012. Dr Su has headed all drug discovery and research since he joined the company, including master-minding the company’s scientific strategy, being a key leader of the Innovation Platform, and responsible for the discovery of each and every small molecule drug candidate in the company’s product pipeline. Prior to joining the company in 2005, Dr Su spent 15 years with the US research and development department of Pfizer, Inc. with his last position as director of the medicinal chemistry department. Dr Su received a bachelor of science degree in chemistry from Fudan University in Shanghai. He completed a PhD and post-doctoral fellowship in chemistry at Harvard University under the guidance of Nobel Laureate Professor EJ Corey. |
Principal shareholders |
(%) |
CK Hutchison Holdings Ltd (through its wholly owned subsidiary Hutchison Healthcare Holdings) |
46.79 |
The Capital Group Companies Inc. |
7.22 |
M&G plc |
4.28 |
Schroders plc |
3.59 |
Fidelity International Ltd |
3.55 |
Invesco Ltd |
2.55 |
BlackRock Inc. |
2.55 |
Mitsui & Co. Ltd |
2.27 |
|
Companies named in this report |
AstraZeneca (LON:AZN), CK Hutchison (SEHK:0001), Nestlé SA (VX:NESN), Guangzhou Baiyunshan (SHA: 600332, SEHK:874), Shanghai Pharmaceuticals (SHA: 601607, SEHK: 2607), Eli Lilly (NYSE:LLY), Novartis (SWX:NOVN), Pfizer (NYSE:PFE), Boehringer Ingelheim |
|
General disclaimer and copyright
This report has been commissioned by Hutchison China MediTech and prepared and issued by Edison, in consideration of a fee payable by Hutchison China MediTech. Edison Investment Research standard fees are £49,500 pa for the production and broad dissemination of a detailed note (Outlook) following by regular (typically quarterly) update notes. Fees are paid upfront in cash without recourse. Edison may seek additional fees for the provision of roadshows and related IR services for the client but does not get remunerated for any investment banking services. We never take payment in stock, options or warrants for any of our services.
Accuracy of content: All information used in the publication of this report has been compiled from publicly available sources that are believed to be reliable, however we do not guarantee the accuracy or completeness of this report and have not sought for this information to be independently verified. Opinions contained in this report represent those of the research department of Edison at the time of publication. Forward-looking information or statements in this report contain information that is based on assumptions, forecasts of future results, estimates of amounts not yet determinable, and therefore involve known and unknown risks, uncertainties and other factors which may cause the actual results, performance or achievements of their subject matter to be materially different from current expectations.
Exclusion of Liability: To the fullest extent allowed by law, Edison shall not be liable for any direct, indirect or consequential losses, loss of profits, damages, costs or expenses incurred or suffered by you arising out or in connection with the access to, use of or reliance on any information contained on this note.
No personalised advice: The information that we provide should not be construed in any manner whatsoever as, personalised advice. Also, the information provided by us should not be construed by any subscriber or prospective subscriber as Edison’s solicitation to effect, or attempt to effect, any transaction in a security. The securities described in the report may not be eligible for sale in all jurisdictions or to certain categories of investors.
Investment in securities mentioned: Edison has a restrictive policy relating to personal dealing and conflicts of interest. Edison Group does not conduct any investment business and, accordingly, does not itself hold any positions in the securities mentioned in this report. However, the respective directors, officers, employees and contractors of Edison may have a position in any or related securities mentioned in this report, subject to Edison's policies on personal dealing and conflicts of interest.
Copyright: Copyright 2020 Edison Investment Research Limited (Edison).
Australia
Edison Investment Research Pty Ltd (Edison AU) is the Australian subsidiary of Edison. Edison AU is a Corporate Authorised Representative (1252501) of Crown Wealth Group Pty Ltd who holds an Australian Financial Services Licence (Number: 494274). This research is issued in Australia by Edison AU and any access to it, is intended only for "wholesale clients" within the meaning of the Corporations Act 2001 of Australia. Any advice given by Edison AU is general advice only and does not take into account your personal circumstances, needs or objectives. You should, before acting on this advice, consider the appropriateness of the advice, having regard to your objectives, financial situation and needs. If our advice relates to the acquisition, or possible acquisition, of a particular financial product you should read any relevant Product Disclosure Statement or like instrument.
New Zealand
The research in this document is intended for New Zealand resident professional financial advisers or brokers (for use in their roles as financial advisers or brokers) and habitual investors who are “wholesale clients” for the purpose of the Financial Advisers Act 2008 (FAA) (as described in sections 5(c) (1)(a), (b) and (c) of the FAA). This is not a solicitation or inducement to buy, sell, subscribe, or underwrite any securities mentioned or in the topic of this document. For the purpose of the FAA, the content of this report is of a general nature, is intended as a source of general information only and is not intended to constitute a recommendation or opinion in relation to acquiring or disposing (including refraining from acquiring or disposing) of securities. The distribution of this document is not a “personalised service” and, to the extent that it contains any financial advice, is intended only as a “class service” provided by Edison within the meaning of the FAA (i.e. without taking into account the particular financial situation or goals of any person). As such, it should not be relied upon in making an investment decision.
United Kingdom
This document is prepared and provided by Edison for information purposes only and should not be construed as an offer or solicitation for investment in any securities mentioned or in the topic of this document. A marketing communication under FCA Rules, this document has not been prepared in accordance with the legal requirements designed to promote the independence of investment research and is not subject to any prohibition on dealing ahead of the dissemination of investment research.
This Communication is being distributed in the United Kingdom and is directed only at (i) persons having professional experience in matters relating to investments, i.e. investment professionals within the meaning of Article 19(5) of the Financial Services and Markets Act 2000 (Financial Promotion) Order 2005, as amended (the "FPO") (ii) high net-worth companies, unincorporated associations or other bodies within the meaning of Article 49 of the FPO and (iii) persons to whom it is otherwise lawful to distribute it. The investment or investment activity to which this document relates is available only to such persons. It is not intended that this document be distributed or passed on, directly or indirectly, to any other class of persons and in any event and under no circumstances should persons of any other description rely on or act upon the contents of this document.
This Communication is being supplied to you solely for your information and may not be reproduced by, further distributed to or published in whole or in part by, any other person.
United States
Edison relies upon the "publishers' exclusion" from the definition of investment adviser under Section 202(a)(11) of the Investment Advisers Act of 1940 and corresponding state securities laws. This report is a bona fide publication of general and regular circulation offering impersonal investment-related advice, not tailored to a specific investment portfolio or the needs of current and/or prospective subscribers. As such, Edison does not offer or provide personal advice and the research provided is for informational purposes only. No mention of a particular security in this report constitutes a recommendation to buy, sell or hold that or any security, or that any particular security, portfolio of securities, transaction or investment strategy is suitable for any specific person.
Frankfurt +49 (0)69 78 8076 960 Schumannstrasse 34b 60325 Frankfurt Germany |
London +44 (0)20 3077 5700 280 High Holborn London, WC1V 7EE United Kingdom |
New York +1 646 653 7026 1,185 Avenue of the Americas 3rd Floor, New York, NY 10036 United States of America |
Sydney +61 (0)2 8249 8342 Level 4, Office 1205 95 Pitt Street, Sydney NSW 2000, Australia |