Transitioning focus to FDA approval for VF-001
Factor Therapeutics (formerly named Tissue Therapies) is developing VF-001, an advanced wound care product designed to accelerate the healing of chronic VLU, diabetic foot ulcers (DFU) and other chronic wounds. VF-001 is a fusion protein that combines an extracellular matrix component (vitronectin) with a growth factor (IGF-1). The fusion protein acts both to restore the extracellular matrix (ECM) and to support the growth and migration of key cells involved in wound healing that are dependent on anchorage. The vitronectin component rapidly binds to collagen in the ulcer bed to localise the growth factor, IGF-1, at the site where regeneration needs to occur.
In 2012 Factor applied for CE mark approval of VF-001 (known then as VitroGro) as a Class III medical device to aid healing in VLU, based on data from a 53-patient open label trial. However, in June 2015 after a lengthy review process that involved several twists and turns, the company received a Final Advice Letter from the European Medicines Agency (EMA) advising that it wanted additional safety and preclinical data. At that point Factor decided to withdraw its application for CE Mark approval. The additional information included further preclinical studies to demonstrate the therapeutic utility of the IGF-1 component of VF-001 and clinical studies to demonstrate the comparative safety of the VF-001 construct.
During the CE mark review process the UK Medicines and Healthcare products Regulatory Agency (MHRA) ruled that VF-001 should be assessed as a Class III (ie high risk) medical device under Rule 13. Rule 13 applies to devices “incorporating…a substance which, if used separately, can be considered to be a medicinal product” such as drug eluting stents and wound dressings incorporating antimicrobial agents. As we understand it, the classification as a medical device is based on the premise that whereas bioengineered skin graft products such as EpiFix or Oasis Wound Matrix consist largely of extracellular matrix proteins formulated as a solid sheet, VF-001 comprises a synthetic ECM protein in solution and acts as a topically applied biomimetic scaffold. Rule 13 applies because VF-001 also contains the IGF-1 growth factor linked to the vitronectin ECM protein.
Separately, in October 2011 the company was advised that the US FDA would classify VF-001 as a biologic product (and not a device). This is considered the usual FDA classification for synthetic protein products used for human treatment. Factor is planning to initiate a randomised Phase IIb trial as part of a strategy to pursue FDA approval of VF-001 as a biologic, initially for the treatment of VLU. Whereas a confirmatory Phase III trial would be required before filing for FDA approval, the Phase IIb trial (if successful) should provide sufficient data to enable the company to reapply for approval as a medical device in Europe.
Venous leg ulcers an initial target indication
VLU are caused by changes in the blood flow in the veins of the legs from blockages and/or weakness in the valves, which result in sustained high pressure in the veins. Up to 1% of adults will develop a VLU at some time in their life. VLU comprise around three-quarters (60-85%) of all leg ulcers. An ulcer is generally considered chronic if it remains unhealed after four weeks. Following healing, there is recurrence in 26-69% of people within 12 months.
The standard treatment for chronic VLU is multilayer compression bandaging and moisture retentive dressings, which are changed weekly. Research has shown that compression improves healing rates compared to treatments using no compression and that high compression achieves better healing rates than low compression.
VF-001 – synthetic matrix protein that promotes wound healing
An important step in wound healing is the formation of a healthy wound bed to which skin cells can attach as they proliferate and migrate onto the wound bed during the healing process. Extracellular matrix proteins are an important component of a healthy wound bed, but are often degraded in chronic wounds.
VF-001 is a synthetic protein designed to promote wound healing. It contains a portion of the ECM protein vitronectin (amino acids 1-64) linked to the growth factor IGF-1 (insulin-like growth factor). The vitronectin component contributes a collagen-binding site that anchors the protein to the wound bed and a cell attachment site that provides a scaffold facilitating the movement of cells onto the wound bed. The IGF-1 portion stimulates proliferation and migration of keratinocytes and fibroblasts needed for new skin formation (Exhibit 1).
Previous clinical studies have shown that treatment with VF-001 can convert non-healing wounds into healing wounds. VF-001 has shown consistent performance in long-standing non-healing ulcers, with one-third completely healed in 12 weeks. VF-001 is applied once per week, so its use fits in well with the recommended weekly change of standard of care compression bandaging combined with moisture retentive dressings.
Exhibit 1: VF-001 combines an extra cellular matrix protein with a growth factor
|

|
Source: Factor Therapeutics website
|
VF-001 is protected by a number of different patents and other market exclusivities. Core patents for VF-001 expire in 2024, while new constructs have patent life out to 2031. However, based on our anticipated 2023/24 approval date in the US, we anticipate that the 12 years of BLA exclusivity in the US will provide protection in that market until 2035. As a biological product with modest forecast peak sales, we do not expect competition from biosimilar products to arrive until several years after market exclusivity expires.
Preclinical studies show that VF-001 accelerates wound healing
In preclinical studies in a porcine wound model, treatment with VF-001 led to faster wound healing than in wounds treated with vehicle control (Exhibit 2). Although these were acute rather than chronic wounds, they give a clear demonstration that VF-001 can accelerate wound healing.
Exhibit 2: VF-001 accelerated healing in a porcine deep partial thickness wound model
|

|
Source: Factor Therapeutics. Data based on Shooter et al 2013, International Wound Journal doi: 10.1111/iwj.12073.
|
VF-001 promoted healing of non-healing ulcers in clinical trial
An open-label Phase II clinical trial (VitroCARD 1001) was conducted as part of a strategy to seek CE mark approval in Europe as a medical device. The 12-week, prospective, multicentre study recruited 53 patients with hard-to-heal chronic VLU that had failed to respond to at least four weeks of standard of care (moist wound healing and compression). This trial was enriched for subjects for whom individualised care at major treatment centres had failed. The average age of patients was 74 years, average ulcer duration was 33 months and average baseline ulcer size was 7.4cm2.
The VF-001 was applied to the wound bed each time the standard moisture-retentive dressing and compression bandaging were changed. In a peer-reviewed report of the study, the authors concluded that VF-001 was “an effective and safe adjunct to standard care in the treatment of hard-to-heal leg ulcers”.
The chronic VLU had completely healed by the end of the 12-week treatment period in 30% of patients who started the study (n=53, the intention to treat data set) and 35% of those who completed treatment (n=45, completers data set). Furthermore, 67% of patients had more than a 50% reduction in wound area. The study found there was no difference in efficacy between once-weekly and twice-weekly treatment with VF-001.
Exhibit 3 shows an example of a response to the treatment. This patient had a large ulcer that had been present for five years, but fully healed after seven weeks of treatment with VF-001 combined with standard compression therapy.
The treatment was safe and well tolerated. There were no adverse events that were considered to be definitely or probably associated with VF-001 therapy. Thirteen adverse events were considered to be possibly related to VF-001. The most common of these was hypergranulation (excess vascular tissue), which is considered to be a sign that wound healing is underway.
Exhibit 3: VF-001 plus compression prompted healing in long-standing ulcers in Phase II trial
|

|
Source: Factor Therapeutics website
|
Comparison to historical control group suggests greatest benefit in VLU of intermediate severity
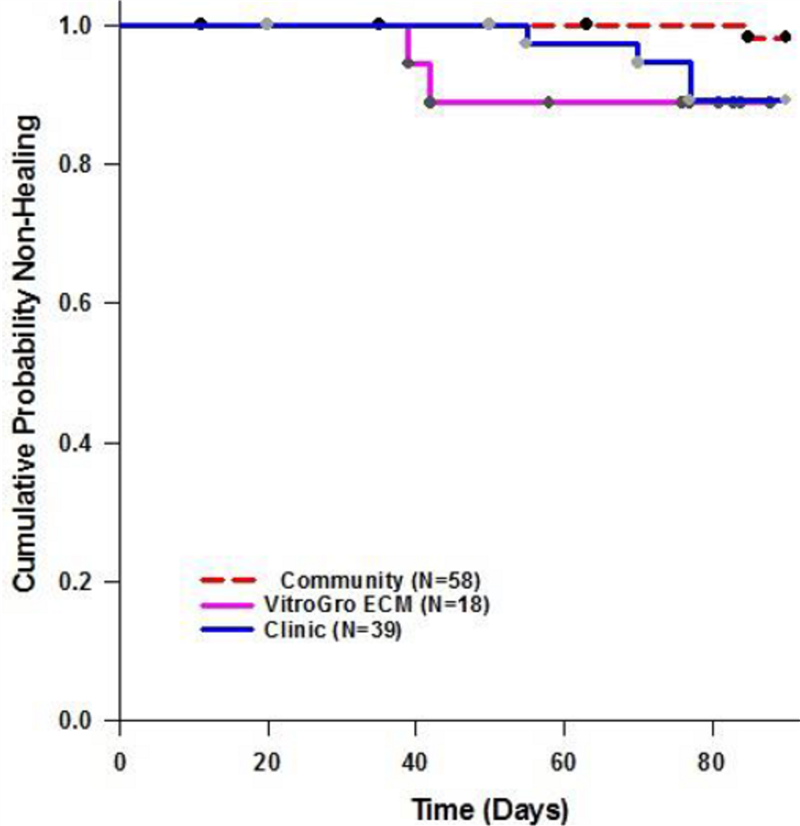
The Phase II trial was an open-label study that did not include a control group for comparison, a design that is in common in medical device studies. To estimate the likely benefit of treating patients with VF-001 in addition to standard care, two historical control groups were established comprising patients matched on the basis of age, ulcer characteristics and treatment history.
The two historical control groups were constructed using the records of patients treated with standard compression bandaging in the VenUS series of multicentre randomised controlled VLU trials managed by the University of York in the UK. Patients from the VenUS data set were matched to patients in the VitroCARD 1001 trial on the basis of patient age, ulcer duration and ulcer area at baseline, factors which have been shown to predict the propensity of ulcers to heal.
Exhibit 4: Patients treated with VF-001 healed faster than propensity-matched historical control group
|
Exhibit 5: VF-001 showed greatest efficacy in “Margolis 1” VLU of intermediate severity
|

|

|
Source: Factor Therapeutics
|
Source: Factor Therapeutics
|
Exhibit 4: Patients treated with VF-001 healed faster than propensity-matched historical control group
|

|
Source: Factor Therapeutics
|
Exhibit 5: VF-001 showed greatest efficacy in “Margolis 1” VLU of intermediate severity
|

|
Source: Factor Therapeutics
|
The main historical control group comprised patients who received weekly treatment in a specialty wound clinic, making them directly comparable to the VF-001 Phase II trial. A second historical control group comprised patients undergoing treatment in a community setting to allow an estimate of the potential benefit of VF-001 when used in a community setting where the majority of VLU patients are actually treated.
While acknowledging that a retrospective comparison to a historical control group has serious limitations, we note that Exhibit 4 shows that VF-001 may provide a tangible benefit in accelerating healing of chronic VLU. Whereas the probability of non-healing at 12 weeks in VitroCARD 1001 trial was ~60%, in the historical control clinic group the probability was ~80%. The company has further investigated the effect of VF-001 on VLU of varying severity. This analysis is based on the work of Margolis et al. (2000) who developed a simple prediction rule to identify patients in whom a VLU is unlikely to heal within 24 weeks (~6 months) if treated with a compression bandage applied weekly.
Margolis assigned one point for an ulcer that is older than six months and another point if it is larger than 5cm2. Patient scores ranged from zero (small ulcer no more than six months old) to two (large ulcer of more than six months’ duration). In the initial analysis of 260 consecutive patients treated at the Cutaneous Ulcer Centre at University of Pennsylvania Medical Centre in 1993-1995, 45% of patients had a Margolis score of zero, 29% had a score of one and 26% had a score of two (Exhibit 6). Exhibit 6 shows that the Margolis score was able to identify which patients would be slow to heal both in the specialty wound clinic population and in a separate population comprising 219 patients in the control arms of two VLU clinical trials.
Exhibit 6: Margolis score predicts which VLU patients will be slow to heal
Margolis severity score4 |
Proportion in clinic population |
Per cent healed within 24 weeks – clinic population* |
Per cent healed within 24 weeks – clinical trial population# |
Baseline ulcer area (cm2) |
Ulcer duration (months) |
0 (least severe) |
45% |
93% |
95% |
≤ 5 |
≤ 6 |
1 (moderate severity) |
29% |
65% |
73% |
≤ 5 |
> 6 |
> 5 |
≤ 6 |
2 (most severe) |
26% |
13% |
37% |
> 5 |
> 6 |
Source: Margolis et al. 20004, Edison Investment Research. Note: *260 consecutive clinic patients; #219 patients enrolled in the standard care (control) arms of two similar randomised VLU clinical trials.
Factor found that in the VitroCARD 1001 trial VF-001 worked best in patients with a Margolis score of 1 (moderate severity). Exhibit 6 (above) shows that subjects with a Margolis score of one who were treated with VF-001 had a probability of healing at 12 weeks of 60%, whereas the probability was ~25% in the specialty wound clinic control group and ~20% in the community care setting historical control group.
Exhibit 7: VF-001 led to earlier healing in Margolis 0 (mild) ulcers compared to historical control group
|
Exhibit 8: Healing of Margolis 2 (severe) ulcers treated with VF-001 similar to historical control group
|

|
|
Source: Factor Therapeutics
|
Source: Factor Therapeutics
|
Exhibit 7: VF-001 led to earlier healing in Margolis 0 (mild) ulcers compared to historical control group
|

|
Source: Factor Therapeutics
|
Exhibit 8: Healing of Margolis 2 (severe) ulcers treated with VF-001 similar to historical control group
|

|
Source: Factor Therapeutics
|
Among subjects with the least severe ulcers (Margolis 0) patients in the VF-001 trial healed faster than those in the historical control group, but by 12 weeks the difference in healing rates was relatively modest (Exhibit 7). In the more severe Margolis 2 patients, only a small number healed in either group (Exhibit 8).
Consistent with this analysis, Factor is targeting development of VF-001 as a treatment for chronic VLU of intermediate severity, most of which are treated in the community setting. It estimates that this will include ~30% of VLU patients.
The Margolis scoring system is not widely used in clinical practice, so the company will not use a “Margolis score” to screen patients for the upcoming trial. Instead it will use other enrolment criteria to enrich the trial for patients with VLU of moderate severity, which would be approximately equivalent to a Margolis 1 score.
Targeted ‘precision’ trial design
Factor is in the final stages of preparing for the Phase IIb trial of VF-001 in chronic VLU, having submitted an amended IND on 7 October that contained manufacturing certifications and a revised clinical trial protocol. This should allow the trial to start recruiting patients in Q416. The full details of the trial will be released when the amended IND is accepted by the FDA.
To enrich the trial for patients with ulcers of intermediate severity, we expect it to include a run-in period of around four weeks of standard compression therapy (this treatment could potentially be administered before recruitment if documentation of wound healing rate is adequate). Patients whose ulcers show signs of healing during the run-in period will be excluded from the trial, which should eliminate the majority of patients with less severe ulcers who would be likely to heal after 12 weeks of standard care regardless of whether they were treated with VF-001.
Additional recruitment criteria will be put in place to reduce the number of patients in the trial with severe ulcers that are unlikely to fully heal within a 12-week treatment period, even with the addition of VF-001.
The company has disclosed that the Phase II will be a multicentre, randomised placebo-controlled study that is anticipated to recruit 170 patients at 26 trial sites. Patients will be randomised 1:1:1 to receive treatment with a placebo, standard or high dose of VF-001. They will receive weekly applications of VF-001 (or placebo) for 12 weeks in addition to standard moist dressings and compression therapy and will be followed for a further 12 weeks after the completion of the treatment period.
The primary endpoint will be the percentage of reduction in ulcer area at the end of treatment. This will allow the study to identify the time when there is the greatest difference between VF-001 and placebo, which could influence the design of Phase III studies.
Secondary endpoints will include the proportion of patients with complete ulcer closure after 12 weeks of treatment, which is the EMA’s preferred efficacy endpoint. Complete ulcer closure would also be the primary endpoint for subsequent US Phase III studies.
Pain reduction and improvement in quality of life will be additional secondary endpoints.
Factor has manufactured and packaged irrigation syringes containing VF-001 at either a standard dose (14ug/mL) or a high dose (140ug/mL) or placebo, for the trial. The trial has been designed to comply with the data gathering requirement outlined by the EMA as part of the company’s withdrawal from the CE marking process in 2015. In conjunction with modest non-clinical activity, the data from the US Phase II could be included in a CE mark resubmission to the EMA.
Competition in advanced wound care
As Exhibit 9 shows, no biological growth factor drugs have been approved specifically for the treatment of chronic VLU, and very few advanced wound care products of any kind have been approved for VLU.
Exhibit 9: Products marketed for VLU or DFU or in development for VLU
Product |
Company |
Indication |
Status |
Description |
Development notes |
Approved for VLU |
|
|
|
|
Apligraf (celtx, graftskin) |
Organogenesis Inc. |
VLU, DFU |
Marketed |
Bi-layered skin substitute: human keratinocytes and human fibroblasts in a bovine collagen lattice |
FDA approved Apligraf for VLU in 1998 and DFU in 2000. Pivotal VLU study in 240 patients: Apligraf group 45% healed at 12 wks vs 38% controls. 24-week data: Apligraf 55% healed vs 49% in controls - Regression analysis significant at 24 weeks p=0.02. |
Oasis Wound Matrix |
|
VLU, DFU and other wounds |
Marketed |
Porcine derived extracellular matrix sheets |
Indicated for the management of chronic and traumatic wounds. 120-pt randomised 12-week trial in chronic ulcers 55% healed in Oasis arm vs 34% control (p=0.02). |
ReGenerCell |
Avita Medical |
VLU, DFU and other wounds |
Marketed EU/ Australia/ China (investigational in the US) |
Autologous suspension of patient's own skin cells. |
CE-mark, TGA cleared, CFDA-cleared in China for treatment of chronic wounds including VLU and DFU. UK 14-wk VLU, trial March 2016; n=53. Wound closure 27% for treated group vs 15% in control - not significant. Investigational use only in the US. |
Approved for DFU but not VLU |
|
|
|
|
Collagenase Santyl |
Smith & Nephew |
Debriding ulcers, burns |
Marketed |
Enzymatic debriding ointment |
Applied daily to aid the removal of dead tissue and allow healing. Necrotic tissue is more common in DFU than VLU. |
|
Regranex (becaplermin) |
Smith & Nephew |
DFU, pressure ulcers |
Marketed |
Recombinant platelet derived growth factor |
In two small, randomised 16-week VLU studies combined incidence of complete closure was 46% (30/65) for Regranex vs 39% (26/67) for control (not significant). Regranex is the only FDA approved biologic drug for DFU. |
|
EpiFix |
MiMedix |
Minimally modified, not requiring premarket approval |
Marketed |
Dehydrated human amnion/chorion membrane allograft |
Outperformed Apligraf in a head to head DFU study. After six weeks 95% of EpiFix patients achieved complete healing compared to only 45% with Apligraf. FDA certified as a Human Cells, Tissues and Cellular and Tissue-Based Product (HCT/P). |
|
Dermagraft (ABH001) |
Organogenesis Inc. |
DFU, pressure ulcers |
Marketed |
Engineered skin substitute containing live human fibroblasts |
366-pt VLU trial failed to meet primary endpoint Aug 2011. At 12 weeks 34% (64/186) healed in Dermagraft group vs 31% (56/180) controls (p=0.24). Patients enrolled if ulcer healed by <50% in two-week screening period. |
|
Omnigraft |
Integra LifeSciences |
DFU, burns |
Marketed |
Sheet of silicone, cow collagen, shark cartilage |
FDA approved for burns in 1996. FDA approved for DFU Jan 2016. |
|
Failed VLU Phase III/ Stalled in Phase II |
|
|
|
|
HP802-247 |
Smith & Nephew |
VLU (failed) |
Failed Phase III |
Spray of a fibrinogen solution and a cell preparation |
US 440-pt PhIII in VLU failed in 2014 -recruited patients with VLU at least six weeks but <2 yrs duration. Europe PhIII terminated Feb 2015. PhII 58-70% in treated groups healed at 12 weeks vs 46% in controls. |
|
CureXcell |
Macrocure Ltd. |
VLU, DFU both failed |
Failed Phase III |
Activated donor white blood cells |
Terminated VLU PhIII for futility Aug 2015. DFU PhIII missed primary endpoint Oct 2015. |
|
Nexagon |
CoDa Therapeutics Inc. |
VLU, DFU |
Phase II (Stalled) |
Antisense oligonucleotide that targets gap junction protein alpha 1 |
Inhibits inflammation. 300-pt Novel2 PhII VLU trial completed Mar 2013. Results described as positive, but no further development. 168-pt DFU PhII completed Apr 2014 - no results announced. |
|
Source: BioCentury, Factor Therapeutics internal company documents, Edison Investment Research
A much larger number of products have been approved for treating DFU, partly because the high potential healthcare cost savings from reducing the incidence of lower limb amputation has enabled higher prices to be charged, and has therefore attracted greater attention from drug developers. The second section of Exhibit 9 lists some of the main products that have been approved for treating DFU but not for treating VLU. One of these products, Dermagraft, failed a pivotal trial in VLU in 2011, while the other products have not been tested in large VLU trials.
The development pipeline for advanced wound care products to treat VLU is very sparse. We have not identified any products that are actively undergoing Phase II or Phase III trials in VLU indication, whereas there are several products in Phase III for DFU. It is possible that one of the products that is currently in development or already approved for treating DFU may undergo subsequent trials in a VLU indication.
Reimbursement criteria for other VLU products in the US consistent with anticipated VF-001 trial design
The guidelines from the health insurer Wellmark in the US give an indication of the conditions that are likely to be needed to be met to get reimbursement for VF-001 in the US. Wellmark reimburses what it terms “bioengineered skin and soft tissue substitutes” Apligraf and Oasis Wound Matrix for chronic venous leg ulcers “which have not adequately responded following a one-month period of conventional ulcer therapy (there has been no improvement of the wound with regular dressing changes and wound care)”.
Given that the screening process for the upcoming Phase II trial of VF-001 is likely to include at least four weeks of trial of compression therapy and to only randomise patients whose ulcers have not shown adequate healing in this period, we believe that VF-001 is likely to be reimbursed according to similar criteria to those applying to Apligraf and Oasis in the US.
VF-001 is formulated as a solution for wound irrigation. We expect the commercial product to be a kit containing sufficient syringes for a course of treatment, eg 12 irrigation syringes of VF-001. If the drug is further developed for other indications such as DFU, it may be reformulated as a gel for ease of application.
Forecasting VF-001 sales potential
Factor is initially targeting approval of VF-001 for the treatment of chronic venous leg ulcers. There are no comparable products approved for this condition. VF-001 is targeting use in a community setting, whereas most advanced wound care products are used primarily in specialised wound care centres. This will allow Factor to target a larger proportion of VLU patients, which has led us to do a bottom-up analysis to identify the number of patients who would be candidates for treatment if Factor’s product is approved.
It is commonly cited that 1% of the adult population suffers from leg ulcers. This figure refers to the combined total of people who currently have an ulcer or who have a history of leg ulcers. However, the figure that is relevant to Factor in identifying the target population is the number of people who will develop a new or recurrent VLU each year.
A number of different approaches have been used to estimate the VLU incidence, resulting in a wide range of estimates cited in the literature. Exhibit 10 summarises VLU incidence estimates from a range of epidemiology studies. Estimates of incidence or prevalence in the total population range from 0.04% to 0.29%. VLU are more common in the elderly; prevalence estimates range from 0.33% to 0.86% in people aged 60 or over and from 0.21% to 1.7% in those aged over 65. Incidence represents the number of patients who develop a VLU each year, while prevalence counts all patients who had a VLU at any time during the year, including ulcers that developed in the preceding year.
A number of the studies in Exhibit 10 have relied on surveys of medical practitioners to identify VLU cases. However, Walker et al. (2002) concluded that the survey approach can miss over 80% of leg ulcer cases, based on a capture/recapture analysis similar to that used to estimate wildlife populations. To overcome this limitation of surveys, we have based our forecasts on the expected number of VLU cases in two large studies that estimated VLU incidence from large databases of medical records, an approach that seems to be more robust and likely to give a good estimate of the number of patients seeking treatment.
These two large studies (Takahashi et al. 2010 and Margolis et al. 2002) reported the incidence and prevalence of VLU in patients over 60 or over 65. We have used the observations from other studies that 10% of VLU cases occur in patients under 60, and 15% of cases occur in patients under 65 to extrapolate these estimates of VLU occurrence in elderly patients to generate estimates of the number of VLU cases in the total population each year.
Exhibit 10: VLU Incidence/prevalence estimates from epidemiology studies and predicted number of VLU cases in the US in 2016 based on each study’s findings
Study |
VLU Incidence/prevalence (%) |
Predicted |
|
|
Population |
Over 60s |
Over 65s |
US cases |
Description/details |
Margolis8 2002 |
|
|
1.16% |
707,000 |
VLU incidence (excludes recurrences within eight-year period) |
Margolis 2002 |
|
|
1.69% |
1,031,000 |
VLU prevalence (includes ulcers that started in the previous year) |
Takahashi7 et al. 2010 |
|
0.86% |
|
681,000 |
VLU incidence from a retrospective cohort study of adults over 60 |
Walker6 et al. 2002 |
0.18% |
|
|
572,000 |
Auckland. Based on capture/recapture incidence. Study included leg ulcers of all types, assume 70% VLU |
Baker9 et al. 1991 |
0.06% |
0.33% |
|
261,000 |
Chronic (at least one-month duration) VLU prevalence |
Cornwall et al. 1986, cited by Baker 1991 |
0.15% |
|
|
473,000 |
VLU prevalence |
Moffatt10 et al. 2004 |
0.04% |
|
0.22% |
131,000 |
VLU prevalence identified over a four-week period. Total leg ulcer prevalence was 0.045%, 78% of ulcers venous (43%) or primarily venous (35%). 55% had their ulcer for over a year |
Nelzen et al. 1996
|
0.29% |
|
|
940,000 |
We calculated VLU prevalence based on their reported 46% venous aetiology. Reported 0.63% incidence of all leg and foot ulcers, including diabetic and arterial, not healed after six weeks. Surveyed ages 50-89 |
Average of Margolis prevalence and Takahashi incidence (US VLU cases) |
0.26% |
|
1.40% |
856,000 |
|
Source: Edison Investment Research. See footnotes for journal article references
This has allowed our estimates to take into account the increase in the average age of the population since the studies were conducted and differences in the proportion of the population aged over 65 in different markets.
Averaging forecast cases in 2016 based on the prevalence estimates of Margolis for over 65s and the incidence estimate of Takahashi for over 60s gives an estimate of 856,000 cases per year in the US. This is equivalent to an incidence of 1.4% in adults aged 65 or over and 0.26% in the total population.
For convenience we have expressed incidence and prevalence estimates in Exhibit 10 as percentages, even though in the source papers the figures are often expressed as cases per thousand or per 100,000 patient years. We note that in some instances subsequent papers have erroneously quoted cases per 1,000 as if they were percentages. For example, whereas Moffatt et al. (2004) reported a VLU prevalence of >8 per 1,000 in patients aged 85 or older (ie 0.8%) based on a survey of medical practitioners in an area of London, Kimmel et al. (2013) subsequently misquoted this prevalence figure as >8% (ie 10x higher than the actual figure).
Exhibit 11 shows our forecast number of VLU cases each year in the major markets. We assume that the product label for VF-001 would be for use in patients who have failed to heal after a certain period (eg, four weeks) of standard compression bandaging. We assume that the addressable market for VF-001 would include 50% of VLU patients, comprising the 29% of patients with ulcers of intermediate severity (Margolis 1) plus half of the patients with the least severe ulcers (Margolis 0). This would be equal to ~425,000 patients in the US each year.
Exhibit 11: Forecast annual number of VLU cases in major markets
Market |
Population (m) |
% aged 65 or over |
Pop'n aged 65 or over (m) |
VLU cases* |
US |
324 |
16.0% |
51.8 |
856,000 |
Eurozone plus UK |
405 |
19.7% |
79.6 |
1,315,000 |
Japan |
127 |
25.7% |
32.6 |
539,000 |
Australia |
24 |
14.7% |
3.5 |
58,000 |
Source: Edison Investment Research, US Census Bureau, World Bank. Note: *Number of VLU cases in each market assumes an incidence of 1.4% in adults aged 65 or over and a further 15% of VLU cases occurring in people aged under 65.
Pharmacoeconomic analysis:
In an analysis of health insurance records of matched pairs of Medicare beneficiaries and privately insured patients in the US, Rice et al (2014a) found that patients with a VLU incurred US$6,710 in additional annual medical costs compared with non-VLU patients. VLU patients covered by Medicare incurred US$6,391 in additional costs (US$18,986 vs US$12,595), whereas privately insured patients incurred an extra US$ 7,030 of costs (US$13,653 vs US$6,623). Privately insured VLU patients missed an extra 4 days from work (14 vs 10), resulting in 29% higher work-loss costs.
In a similar analysis looking at DFUs, Rice et al (2014b) found that patients with a DFU incurred an average of US$14,300 in additional annual health care costs compared to matched controls that did not have a DFU (US$11,710 for Medicare patients and US$16,883 for private insurance).
The additional costs incurred by patients with VLUs means there is considerable scope for cost savings from a treatment that leads to faster healing of VLUs. Factor has conducted pharmacoeconomic analyses to estimate the costs and benefits of treating VLU patients with VF-001 in addition to standard care in both the specialty wound clinic and the community care settings. The analysis was based on comparison to the propensity matched historical control groups comprising patients matched on the basis of age, ulcer characteristics and treatment history, as described on page 6 and shown in Exhibit 4.
Time to healing for ulcers that were not healed at the end of the 12-week trial with VF-001 were projected based on the ulcer size and healing trajectory at the end of the trial, assuming that they continued to receive standard care after the trial concluded. The projected mean time to healing was 176 days for participants in the VF-001 trial, compared to 252 days for patients who received standard care at a specialist wound centre. The pharmacoeconomic analysis found that the faster ulcer healing time reduced total annual healthcare costs (eg bandages and clinic visits) by £1,084 (US$1,390) per patient. A similar comparison to an historical control group who were treated in a community setting the cost saving was by £914 (US$1,170) per patient.
This analysis considers only the direct healthcare costs and does not include the improved quality of life for the patients due to reduced pain and faster healing following VF-001 treatment. This additional benefit to patients helps to offset the uncertainty in the estimates due to the fact that the estimated improvements in healing times are based on a retrospective comparison to a historical control group. The upcoming Phase II trial will directly compare VF-001 to standard care and will allow more precise estimates of the cost savings and improvements in quality of life due to VF-001 treatment, which will influence eventual product pricing.
Management has indicated that product pricing for use in the community setting is likely to be negotiated before product launch based on a revised pharmacoeconomic model that incorporates US data including efficacy and quality of life measures from the upcoming Phase IIb trial. Reimbursement for primary care may require specific submissions to enter a “positive list” of products with proven cost effectiveness that are reimbursed for wound care.
Product pricing assumption
Smith and Nephew markets Regranex, the only biologic drug approved by the FDA to improve wound healing in DFU, which is the closest comparator to VF-001 that is on the market. The company also markets Santyl, which is an enzymatic debriding agent for chronic wounds.
According to Bloomberg prescription data, the average price per prescription for Santyl in H116 was US$400, while for Regranex it was US$1,200. In a 2015 study of Apligraf and EpiFix in DFU patients Zelen et al. reported that the average graft product cost per patient was US$1,669/patient for EpiFix (2.15 grafts at US$776 each) and US$9,216/patient for Apligraf (6.2 grafts at US$1,486 each).
In or forecasts we assume that in order to promote strong uptake in VLU patients treated in the community setting a course of treatment with VF-001 will be priced at US$1,000 per patient. The pharmacoeconomic analysis described above indicates that at this price point the use of VF-001 would result in cost savings to the healthcare system. The price represents a 17% discount to Regranex pricing and is 40% less than the cost of a course of EpiFix in the US.
We will review our product pricing assumptions when we have data from the randomised Phase II trial of the extent to which VF-001 speeds healing, and therefore of the number of weeks of compression bandaging that will be avoided by treating patients with VF-001.
Market sales of comparable products
Smith and Nephew reported that global sales in 2015 for its portfolio of advanced wound bioactives totalled $344m. Regranex is the third biggest-selling product in this category for the company, behind Santyl and Oasis Wound Matrix. If Regranex contributed 20% of global sales of wound bioactives then that would represent global sales for the product of US$69m.
According to Bloomberg prescription data the US sales for Regranex and Santyl in 2015 were US$41m and US$107m respectively.
Incidence of diabetes: Second targeted indication
The company plans to file a separate IND to allow the development of VF-001 for the treatment of DFU. We have also performed a bottom-up analysis to identify the number of cases of DFU in the major markets.
According to the Centres for Disease Control and Prevention, in 2014 there were 22m people in the US diagnosed with diabetes (including 0.2m under age of 18), and a further 8m with undiagnosed diabetes. However, we use the number of diagnosed diabetics to calculate the number of DFU cases each year because the prevalence surveys were done in the diagnosed diabetic population.
In a 2006 review of the epidemiology of diabetic foot problems Rieber and McFarland reported that among seven population-based studies the annual DFU incidence ranged from 1% to 4.1% depending on the risk profile of the population. The average annual incidence of DFU in people with diabetes in the seven studies was 2.4%.
Based on an annual incidence of DFU among diagnosed diabetics of 2.4%, and a population of 22 million diagnosed diabetics, we estimate that there are a total of 528,000 DFU cases in the US each year.












