Phase IIb SAPHIR 2 strategy involves two studies
The protocol for the Phase IIb SAPHIR 2 European trial has been completed, whereas the design of the US trial is the preliminary stage (both shown in Exhibit 4). Time wise, the studies should significantly overlap. The US study will have a longer treatment period and could potentially benefit from an initial titration phase of the European study. The focus of the European trial will be on PQ912’s synaptic effects with tools investigated in the Phase IIa trial. Classic Cogstate (working memory/ attention composite) will be used as a primary endpoint to assess PQ912’s efficacy in the European study, while secondary and exploratory endpoints will build on those used in the Phase IIa trial, where several of the tests showed statistically significant or trending changes. In the US trial patients will be treated for longer, so the focus will be on the cognitive effects across domains and daily function measured by the classic CDR-SB.
In forthcoming Phase IIb studies to address any safety issues (see SAPHIR Phase IIa data summary, below), Probiodrug will explore lower doses and a gradual titration to the maximum individually tolerable levels. Dose reduction is feasible because the Phase I trial target showed occupancy was 90% at a dose of 800mg bid, while a dose range of 300-600mg bid would still achieve target occupancy in the range of 70-80% plus. In the European study, Probiodrug will enrol patients into an initial 12-week treatment regimen titrating the dose of PQ912 in the range 150-300mg over that period. Subsequently, patients will receive the maximum individually tolerable dose of 300mg or 600mg. In total, each patient is expected to be treated for between 36 and 84 weeks (on average around 52 weeks). The US trial should be similar in design, but will treat the patients longer for 18 months on average.
Management indicated that the European Phase IIb trial could start in Q418 and final data are expected in Q321. Details of the US study are yet to be finalised, but potentially could recruit the first patient in Q219 with data readout in Q322, so both trials should run substantially in parallel. The rational for this Phase IIb strategy is based on stepwise risk reduction and value creation, the ability to engage with the regulatory authorities to discuss the Phase IIb data sooner and understand in a timely manner whether the data are supportive of early NDA filing with the expectation of conditional approval or whether a Phase III trial will be needed. When it comes to funding, Probiodrug indicated the total cost of the European trial is estimated at €25m and >€60m for the US study. The company could potentially receive funding of up to $15m from the NIH, which would substantially alleviate the burden. There are several options when it comes to funding the trials, including a fund-raise or a partnership.
Exhibit 4: Phase IIb strategy (SAPHIR 2) in Europe and US
European study |
Focus on synaptic effects with tools investigated in Phase IIa |
Summary design |
Prospective, multi-centre, randomised controlled trial |
Design details |
250 early-stage AD patients 12 weeks of treatment with 150-300mg (bid) for initial safety readout Patients then receive individually highest tolerated doses Each patient will be treated for 36 to 84 weeks (mean 12 months) |
Patients |
Male or female; MMSE 21-30, CSF amyloid & tau positive; on standard of care or treatment naïve |
Endpoints |
Primary: working memory/attention composite (Cogstate) Secondary: effect on qEEG on synaptic function and brain connectivity, total Cogstate and Instrumental Activities of Daily Living (IADL) Exploratory readouts: CSF-based biomarker and MRI imaging of brain and hippocampal volume |
Timelines |
Trial start in coming months, depending on funding; recruitment duration 16 months; |
|
|
US study |
Focus on cognitive effects across domains and daily function |
Summary design |
Prospective, multi-centre, randomised controlled trial |
Design details |
460 early-stage AD patients At least 8 weeks of treatment with 150–300mg (bid) for initial safety readout in first 180 patients Patients then receive individually highest tolerated doses Long-term drug exposure, 18 months on average |
Patients |
Male or female; FDA stage 3+4 (MMSE: 21-30 inclusive, CSF AD pathophysiology amyloid + and pTau & tau/A-beta ratio + Patients on SoC or treatment naïve) |
Endpoints |
Primary: CDR-SB Secondary: efficacy on composite measure of cognition and function (CFC2) Exploratory readouts: CSF and plasma based biomarkers, MRI imaging, EEG network connectivity |
Timelines |
Trial start in 2019, depending on funding |
Source: Probiodrug, Note: MMSE = Mini-Mental State Examination.
SAPHIR Phase IIa data summary
In December 2015, Probiodrug announced the publication of the full Phase I results in a peer-reviewed journal describing first-in-man PQ912 safety and PK/PD, which we reviewed in our previous outlook report. The first data from the Phase IIa SAPHIR trial were announced in June 2017. As a reminder:
■
The Phase IIa SAPHIR study was a safety/tolerability trial, but secondary endpoints included exploratory efficacy tests, such as the neuropsychological test battery (CogState) as cognitive composite, quantitative EEG, resting state functional MRI and a set of molecular biomarkers in the spinal fluid.
■
The highest dose of 800mg bid, tested and well tolerated in the Phase I study, was selected and administered for three months. Probiodrug indicated that this high dose (although not established as maximum tolerated dose over the 7-11 days of treatment in the elderly in Phase I) was strategically selected to:
•
firstly, understand the picture of safety/tolerability in AD patients treated for three months and comply with the EMA guidelines requesting that in Phase I or Phase IIa a maximum tolerated dose should be established; and
•
secondly, to obtain early signs of efficacy over such a short treatment period.
■
The number of patients experiencing adverse events did not significantly differ between the placebo and PQ912 arms (PQ912 n=49, placebo n=45), but the total number of patients who were non-adherent to treatment for any reason was higher in the active arm (PQ912 n=26; placebo n=2; p<0.01). Skin and gastrointestinal (GI) side effects were more common in the PQ912 arm.
■
Despite the short treatment period, several exploratory efficacy endpoints provided statistically significant results or trends pointing to a positive overall picture of the dataset, in our view (discussed in our previous report).
Overall, no major safety concerns associated with PQ912 were established in the Phase IIa trial. The observed skin and GI side effects were manageable, appeared early in the trial and resolved on discontinuation of PQ912.
Long quest for AD medicine
Multiple failures of late stage clinical trials with antibodies targeting Abeta over the last decade have dampened the enthusiasm of investors. The most recent additions to this trend have been BACE inhibitors, with three key late-stage failures in 2018 alone. In February, Merck announced that it was terminating its BACE inhibitor verubecestat Phase III APECS study in prodromal AD. This decision was based on the recommendation from an independent data monitoring committee that if the study continued it would be unlikely that a positive benefit/risk ratio would be established. The results were later published, which showed that despite a large reduction in CSF Abeta, patients in the treatment group declined at a similar rate to the placebo group.
In May, Janssen (part of Johnson & Johnson) announced that it was stopping its Phase IIb/III study in late-onset, preclinical-stage AD and Phase II long-term safety study due to liver toxicity. Also, in June, Eli Lilly and AstraZeneca announced that they were terminating the Phase III AMARANTH and DAYBREAK-ALZ trials for lanabecestat in MCI due to AD or early AD and mild AD dementia. This decision was based on the recommendation from an independent data monitoring committee that if the study continued it would likely not meet the efficacy endpoint.
Current consensus is that therapies should target the earliest stages of the Abeta pathogenic sequence – in pathological terms, ideally before plaques are established, and in clinical terms when there remains potential for a sufficient cognitive reserve.3 This is also reflected in the recently updated EMA and FDA 2018 clinical trial guidelines, which now recommend that clinical trials study an earlier AD population including ‘preclinical’ or non-symptomatic.
One of the likely reasons for lack of efficacy in the previous AD trials is that the stage of AD studied is still too late to slow or stop Abeta formation and toxicity. This means that even if these drugs have prevented further formation of Abeta products, it might still be too late to prevent Abeta toxicity caused by Abeta products that have already formed. An interesting approach was taken by Novartis, which is conducting late-stage prevention trials in asymptomatic patients for its BACE inhibitor CNP520 and its Abeta vaccine CAD106 (Exhibit 5). Patients are selected for these trials based on their ApoE4 genotype. According to clinicaltrials.gov, these trials are due to read out in 2024.
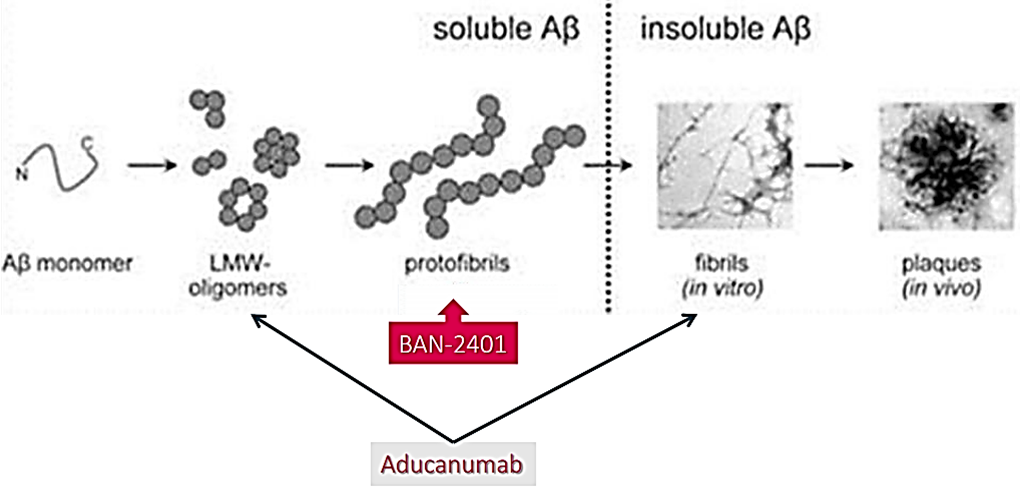
Abeta monoclonal antibodies currently in Phase III trials include aducanumab (Biogen, Eisai) and gantenerumab (Roche), which both have high affinity for aggregated Abeta, and crenezumab (Roche), which has high affinity for oligomeric and fibrillary Abeta and amyloid plaques. These drugs are all targeting early AD. Aducanumab Phase III data are expected in 2020, while multiple studies with gantenerumab and crenezumab should report data over 2020-23 and 2021-23 respectively.
Exhibit 5: Ongoing Abeta Phase III trials
Pharmacological class |
Drug |
Target/mechanism of action |
Company |
Ongoing Phase III trials |
Stage of AD |
Estimated trial completion |
Abeta MAbs |
Aducanumab |
Aggregated Abeta |
Biogen, Eisai |
NCT02477800 |
MCI/mild AD |
2020 |
NCT02484547 |
MCI/mild AD |
2020 |
Gantenerumab |
Aggregated Abeta |
Roche |
NCT02051608 |
Mild AD |
2020 |
NCT03444870 |
Prodromal/mild AD |
2022 |
NCT03443973 |
Prodromal/mild AD |
2022 |
Crenezumab |
Oligomeric and fibrillary Abeta and amyloid plaques |
Roche |
NCT02670083 (CREAD Study) |
Prodromal/mild AD |
2020 |
NCT03114657 (CREAD 2) |
Prodromal/mild AD |
2021 |
NCT03491150 (CREAD OLE open label extension study) |
Prodromal/mild AD |
2023 |
Abeta vaccine |
CAD106 |
Vaccine induces Abeta antibodies which target Abeta N-terminus 1-6 |
Novartis |
NCT02565511 |
At risk of AD* |
2024 |
Small molecule BACE inhibitor |
CNP520 |
BACE1 |
Novartis, Amgen |
NCT03131453 |
At risk of AD* |
2024 |
NCT02565511 (CNP520 and CAD106 combination study) |
At risk of AD* |
2024 |
Elenbecestat |
BACE1 |
Biogen, Eisai |
NCT02956486 (MissionAD1) |
MCI/mild AD |
2020 |
NCT03036280 (MissionAD2) |
MCI/mild AD |
2020 |
Source: clinicaltrials.gov, Global Data, Evaluate Pharma. Note: *Based on age, APOE genotype and elevated amyloid. BACE = Beta-secretase, Abeta = amyloid beta, MAb = monoclonal antibody.