Sports medicine: A large and growing market
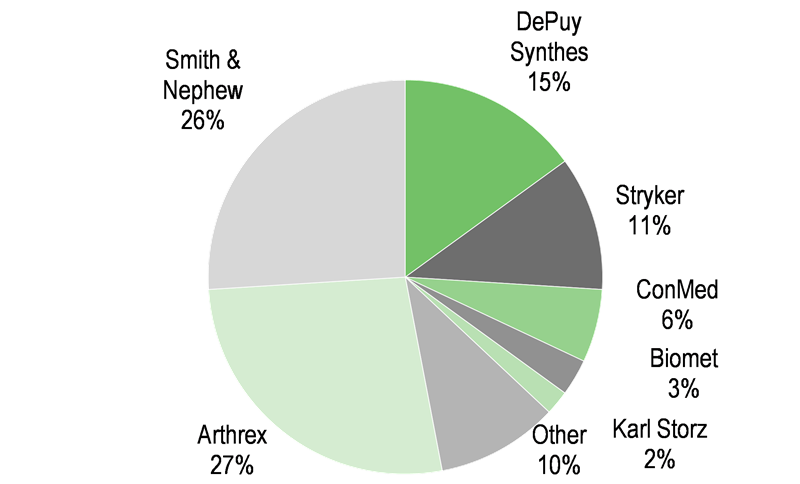
The global orthopaedics market is reportedly worth c $45.5bn and is growing at 3-5% pa (MHBK/IRD). The major segments include joint reconstruction (hip, knee and extremities), spine, trauma, orthobiologics and arthroscopy/soft tissue repair (see Exhibit 7). TRX’s orthopaedic division is involved in two market segments totalling 20% of the overall orthopaedic market: sports medicine (arthroscopy and soft tissue repair) and orthobiologics markets. Both segments are growth leaders in the orthopaedic market (see Exhibit 8).
Exhibit 7: Orthopaedic product sales by market segment
|
Exhibit 8: Orthopaedics market size and growth rates, 2014
|

|

|
Source: MHBK/IRD based on data from ORTHOWORLD
|
Source: MHBK/IRD based on data from ORTHOWORLD
|
Exhibit 7: Orthopaedic product sales by market segment
|

|
Source: MHBK/IRD based on data from ORTHOWORLD
|
Exhibit 8: Orthopaedics market size and growth rates, 2014
|

|
Source: MHBK/IRD based on data from ORTHOWORLD
|
Sports medicine encompasses the therapeutic intervention for injuries or illness resulting from athletic or recreational activities. Orthobiologics are substances that orthopaedic surgeons use to help injuries heal more quickly. They are used to improve the healing of broken bones and injured muscles, tendons and ligaments.
The orthopaedic and sports medicine market has strong growth prospects primarily driven by demographics, in particular the ageing population and the drive to extend active lifestyles. Alongside this is the recognition within the surgical community of the need for a minimally invasive approach and the health economic awareness of the need to do more for less while maintaining quality and function.
The sports medicine market can be broken down into joint repair products (shoulder, knee and hip), which is 46% of the market (see Exhibit 9) and reportedly worth $2.1bn, growing at 7-14%, and arthroscopic enabling technologies, worth $2.5bn growing at 3-6%. The market segment that Tissue Regenix is focused on is joint repair, which comprises products targeting repair of cartilage, ligament and tendons within the joints. These products encompass instruments, orthobiologics, sutures and scaffolds. TRX is focused in the orthobiologics and scaffold space.
Exhibit 9: Global sports medicine market by value (US$)
|

|
|
|
Tissue Regenix is initially focused on two key market segments: meniscal repair and ligament reconstruction, initially anterior cruciate ligaments (ACL) repairs. Meniscal surgery is by far the most common orthopaedic operation, with over 600,000 operations in Europe and nearly one million in the US in 2014 (MRG estimates). Ligament repair surgery is most common in the knee, the anterior and posterior cruciate ligaments (ACL and PCL), with c 750k operations undertaken each year. The majority of those are ACL repairs, with the difference in incidence being 1.8 per 100,000 (PCL injuries) vs 68.6 per 100,000 (ACL injuries).
Meniscal repair: Replacement rather than removal
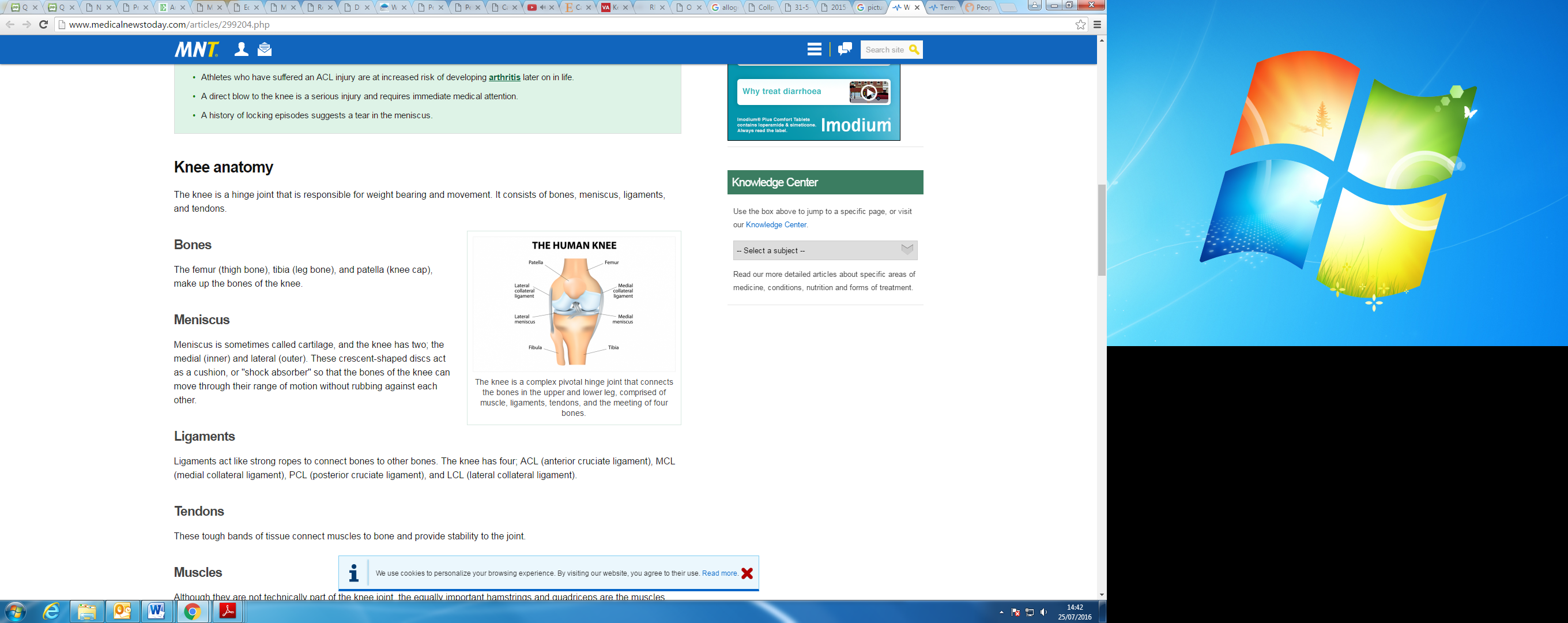
The meniscus is a fibrocartilage structure found within the knee joint (Exhibit 10), and its functions include joint stabilisation, shock absorption and load transmission. The predisposition to injury relates to the limited vascularisation of the meniscus, which is limited in adulthood (c 20% level of vascularisation). It flows radially from the perimeniscal capillary plexus forming three zones: red, red-white and white. The different zones relate to the level of vascularisation, ie the red zone is vascularised, the white avascular and the red-white zone being the transition zone (see Exhibit 11). The level of vascularisation directly correlates to the healing potential of each zone within the meniscus. Damage to the non-vascularised areas of the meniscus, either through tears or degenerative processes, can lead to loss of function of the meniscus. Therefore, tribological function, ie principles of friction, lubrication and wear and stability of the knee joint are compromised6,7.
Exhibit 10: Knee joint, illustrating the anatomical position of the menisci
|
Exhibit 11: Semilunar meniscus, vascular regional variations
|
|

|
|
|
Source: Edison Investment Research
|
Exhibit 10: Knee joint, illustrating the anatomical position of the menisci
|

|
|
|
Exhibit 11: Semilunar meniscus, vascular regional variations
|

|
Source: Edison Investment Research
|
Damage to the meniscus can occur due to disease, degeneration (osteoarthritis), traumatic injury, or abnormal development. Equally, following meniscus injury the patient is predisposed to the development of osteoarthritis. Treatment options can be categorised into removal, repair and replacement (full/partial). They are, however, limited with only 10-20% of meniscal tears being suitable for repair and the remaining 80-90% requiring the removal of part or the whole meniscus. Any meniscal repair solution needs to be able to restore the load-bearing and shock-absorbing functions of the knee, reduce pain and ultimately reduce the long-term need for knee replacement.
Exhibit 12: Available treatments for meniscus surgery
Procedure |
Market share |
Description |
Surgical meniscal repair |
43% of repairs in US,
8% in EU |
<20% of tears are amenable to surgical repair requiring sutures, reabsorbable tacks and more expensive 'hybrid' devices |
Full/partial meniscectomy |
56% in US, 92% in EU |
Risk of osteoarthritis and need for partial or total knee reconstruction later on |
Allograft (following full meniscectomy) |
0.2% (US only) |
From human cadavers, high cost, requires size matching, limited supply, risk of infection/rejection |
Source: MRG data, company websites
Total meniscectomy was, until recently, widely done to treat meniscus injuries. However, research indicated that this type of procedure caused a narrowing of the joint space, lowered rates of regeneration and caused degeneration in the surrounding cartilage and therefore predisposed to osteoarthritis. As a result, a partial meniscectomy is more often performed. Partial meniscectomy involves debriding any meniscal tissue that no longer serves the normal joint function. The amount of meniscal tissue removed has been directly correlated with a decrease in function of the tissue and therefore acts to only alleviate symptoms briefly until the onset of osteoarthritis.
It is clearly preferable, therefore, to repair a meniscus when possible rather than remove via a partial meniscectomy. The meniscus does have an inherent ability to heal itself, although this is limited to the vascular region in its periphery – the red zone. Healing is poor in the innermost zone where there is no blood supply or source of reparative cells (see Exhibit 11 above). Various repair techniques are used, ranging from arthroscopic to open surgery and using sutures or an alternative such as a meniscus arrow, dart, T-fix suture bar or a meniscal screw, although these are not often used now. The predominant approach is arthroscopic suture repair.
Replacement is more favourable than repair as it has a greater potential to protect the joint surfaces. Replacement scaffolds, at varying stages of development, can be autologous (graft taken from the recipient), allogeneic (grafts taken from cadaveric donors) tissue, xenogeneic (animal grafts) or synthetic.
■
Autologous works well and has demonstrated good results. The disadvantages include donor site morbidity, size restriction and cosmetic issues from harvest sites.
■
Allogeneic is the gold standard in meniscal replacement and is available in different forms such as fresh, frozen, lyophilised and cryopreserved. Studies have shown a reduction of symptoms; however, there are problems with the approach including immune rejection, disease transmission and the limitation of donor tissue availability. However, allogenic does have advantages as there is no donor site morbidity, no size restriction, no cosmetic issues from harvest sites and surgery time is reduced as there is no need to harvest from the recipient.
■
Xenogeneic transplantation has become a popular focus, due to its unlimited availability. In terms of size, porcine menisci are the closest match to human menisci. There remains a potential issue of immune rejection, reportedly due to the galactosyl-alpha (1,3) galactose (alpha-gal) epitope present on cell membrane glycolipids and glycoprotein; humans naturally produce high amounts of antibodies to alpha-gal thereby causing the hyperacute rejection.
■
Synthetic materials are an attractive option as they are easily processed, offer minimal batch to batch variability and their mechanical and chemical properties can be tailored. The downside is they can lack the signalling cues present in naturally derived materials for cell repopulation and cause inflammation.
The replacement needs to conform to the biological and biomechanical characteristics of native menisci and importantly integrate, thereby preventing degeneration of the joint. Currently, the two predominant strategies for engineering a meniscal replacement (allogenic, xenographic or synthetic) is either a cell-based method (scaffolds are seeded with cells before implantation), or cell–free method (acellular scaffolds designed to promote and support regeneration once implanted through the infiltration of endogenous cells from the surrounding tissue). The latter offers a shorter translation to clinic timeframe and a better health economic outcome as the former would require harvesting of autologous cells, necessitating two interventions and potential donor site morbidity to produce a personalised implant. It would also require subsequent culture expansion of the cells to obtain sufficient numbers, and subsequent manipulation of cells in culture or a bioreactor system, inevitably being a much higher cost approach.
Attributes which a replacement scaffold requires include:
■
they should be biodegradable and biocompatible – the scaffolds must be able to be absorbed by the tissue;
■
allow ingrowth of vasculature;
■
should not promote an inflammatory or immune response in the tissue;
■
the scaffold surface and porosity should permit cell adhesion and growth and permit the expression of the appropriate cellular phenotype;
■
the material should have the mechanical properties required to withstand the biological demands made on it during the reparative period;
■
the material must be able to be produced reproducibly;
■
scaffold must be able to be supplied sterile in an appropriate size for surgical placement; and
■
the scaffold should have good handling properties, ie able to be trimmed to fit defects and be amenable to manipulation with surgical instruments.